Just Accepted
Just Accepted Articles have been posted online after technical editing and typesetting for immediate view. The final edited version with page numbers will appear in the Current Issue soon.
Submit a Manuscript

Anions-mediated interfacial-water regulation boosts C2+ selectivity for CO2 electroreduction over Cu-based catalysts
Tingting Gong, Zhen Liu, Wen Jin, Jiamei Li, Min Wang*, Luhua Jiang*
https://doi.org/10.1016/j.cjsc.2026.100967
CO2 electrochemical reduction; Cu-based catalysts; Interfacial water; Anions; In-situ ATR-SEIRAS
ABSTRACT
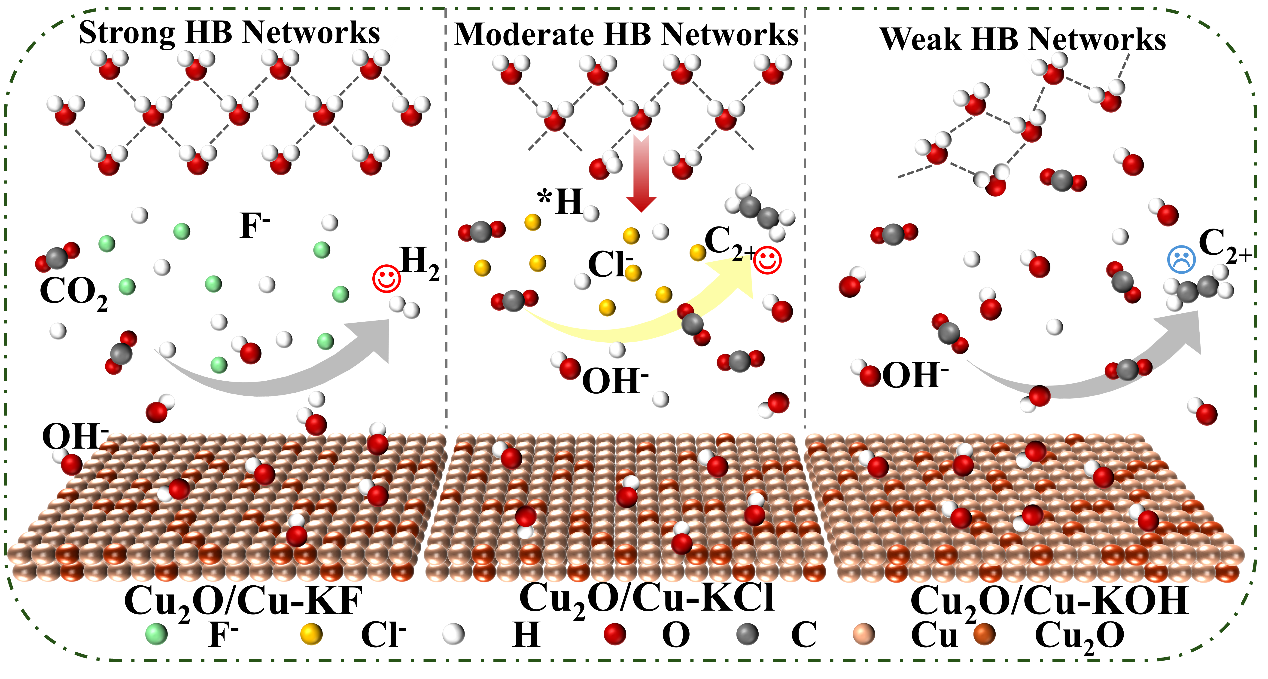
The electrochemical CO2 reduction reaction (CO2RR) offers a promising strategy to produce high-value-added C2+ products. Cu-based catalysts enable C-C coupling but suffer from low C2+ selectivity. Although electronic/structural engineering and cation effects have been widely studied, the role of anions in tuning interfacial water structure and CO2RR pathways remains insufficiently understood. Herein, a series of low-concentration anions were introduced into 1 M KOH to regulate the interfacial water environment over reconstructed CuO. By combining in-situ attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) and electrochemical impedance spectroscopy, it is revealed that low-concentration anions in 1 M KOH increase the proportion of strong interfacial-water structure in the order of F- > Cl- > Br- > NO3-. Moreover, the moderate strong interfacial-water structure can accelerate *H transfer and promote *CO hydrogenation to *COH, thus favoring asymmetric C-C coupling to produce C2+ products. However, excessively stable interfacial water structure in F--containing electrolyte induces enhanced *H coverage and dominant H2 evolution. A volcano-type relationship is thus observed between C2+ selectivity and the proportion of strongly hydrogen-bonded water. This work emphasizes anion-mediated interfacial water regulation as an effective strategy to enhance C2+ selectivity for CO2RR.